 Mortalità americana, dati dal CDC: https://www.cdc.gov/nchs/nvss/vsrr/covid19/excess_deaths.htm#dashboard
Mortalità americana, dati dal CDC: https://www.cdc.gov/nchs/nvss/vsrr/covid19/excess_deaths.htm#dashboard 5-12-20:
Un articolo interessante sulla mortalità della popolazione americana. Per la serie: come si può credere ciecamente alla scienza, quando nemmeno tra scenziati sono d'accordo tra di loro?
Qui c'era un articolo, prontamente ripreso da quei pagliacci di Comedonschisciotte (mi hanno bloccato dopo qualche commento non gradito, alla faccia delle libertà 'alternative'), dove una statistica che pure è ben referenziata, affermava che il COVID non avesse poi ucciso così tante persone, e che la mortalità ad esso attribuita era effettivamente dovuta al ricalcolo di altre patologie, come malattie di cuore e diabete. Abbiamo già visto nel tiggì precedente, che in effetti molte persone sono morte per i rallentamenti nelle terapie che seguivano. Succede anche da noi in Italia, succede in UK, certo succede a maggior ragione negli USA, dove già il sistema sanitario è così inefficiente e costoso, servendo più gli interessi dei privati che quegli ospedali pubblici visti come 'comunisti' da metà della popolazione americana, inclusi quelli che non possono permettersi di essere repubblicani...
OK, quest'articolo è stato ritratto, alla faccia anche dei donchisciottiani.
www.jhunewsletter.com/article/2020/11/a-closer-look-at-u-s-deaths-due-to-covid-19
La ragione per cui in questa newsletter la cosa è stata fatta, dopo appena 4 giorni, la spiegano loro stessi.
A closer look at U.S. deaths due to COVID-19
By YANNI GU | November 27, 2020
Nota dell’editore: Dopo la pubblicazione dell’articolo da parte di The News-Letter il giorno 22 novembre, ci è stato segnalato che il nostro resoconto della presentazione di Genevieve Brians intitolata “Morti da Covid-19: Uno sguardo ai dati statunitensi,” è stato usato per supportare pericolose inesattezze volte a minimizzare l’impatto della pandemia.
Abbiamo deciso, il giorno 26 novembre, di ritirare questo articolo per fermare la diffusione della disinformazione sui social media. Comunque è nostra responsabilità, in quanto giornalisti, lasciare una testimonianza per i posteri. Abbiamo quindi deciso di eliminare l’articolo dal nostro sito, ma di renderlo disponibile a questo indirizzo in formato PDF.
In accordo con i nostri principi di trasparenza, vogliamo condividere con i nostri lettori i motivi che ci hanno portato a questa decisione. The News-Letter è una pubblicazione indipendente sia editorialmente che finanziariamente e gestita dagli studenti. Gli articoli che pubblica non sono avvallati dall’università o dalla scuola di medicina, e la decisione di rimuovere l’articolo è stata presa in modo indipendente.
Lo studio della Briand non deve essere utilizzato come unico mezzo per capire l’impatto del COVID-19, ma deve essere contestualizzato alle innumerevoli altre fonti di dati pubblicate da Hopkins, dall’Organizzazione Mondiale della Sanità e dal Centro per il controllo delle malattie infettive (CDC).
In quanto assistente direttore dei programmi del master di economia applicata all’università Hopkins, Briand non è ne un medico professionista ne un ricercatore medico. Al suo seminario, lei stessa ha sottolineato che sono necessari un numero di dati maggiore e una ricerca più approfondita per quantificare con precisione l’impatto di COVID-19 sui decessi negli Stati Uniti.
Nell’articolo sono state citate queste parole di Briand: “Stando a questi numeri non si può provare che COVID-19 abbia provocato decessi aggiuntivi. Il totale delle morti [durante il periodo in esame] non supera il numero consueto.” Questa affermazione è sbagliata e non tiene in considerazione il picco di decessi derivante da tutte le cause verificatosi rispetto agli anni precedenti. Secondo il CDC ci sono state 300’000 morti aggiuntive causate da COVID-19. Inoltre
Briand ha presentato il confronto fra il totale delle morti negli USA ed il totale dei morti da COVID-19 sotto forma di proporzione percentuale, questo banalizza le ripercussioni della pandemia. Le argomentazioni presentate non confutano la severità del COVID-19; un aumento aggiuntivo non traspare dalle proporzioni perché sono presentate come percentuali e non come semplici numeri.
Nella sua analisi, Briand ha anche affermato che le morti causate da malattie cardiovascolari, malattie respiratorie, influenza e polmonite possono essere state riclassificate erroneamente come dovute a COVID-19. Ma, siccome COVID-19 colpisce soprattutto le persone con malattie preesistenti, è statisticamente molto più probabile che persone con le suddette malattie possano contrarre il virus e morire a causa di esso.
A causa di queste inesattezze e la nostra impossibilità di fornire ulteriori informazioni sull’effetto del COVID-19, The News-Letter ha deciso di ritirare questo articolo. E’ nostro dovere, come giornale, combattere la diffusione della disinformazione e migliorare il nostro processo di verifica. Ci scusiamo con i nostri lettori.
Insomma, una sconfessione su tutta a' ligna. Notare come l'articolo di per se non è considerato scorretto, ma supporta delle pericolose percezioni complottare e negazioniste: ma allora è o non è corretto? Non si può gettare addosso agli 'altri che non capiscono', l'articolo se è corretto dovrebbe essere OK, ma se non lo è, non dovrebbe essere giustificata nemmeno questa ricercatrice. In entrambi i casi, la scienza non ne esce bene: censurare un lavoro di per sé corretto non va bene, pubblicare un lavoro scorretto che dà adito a delle conclusioni scorrette, non va bene ugualmente.
Io penso che sia più la seconda della prima.
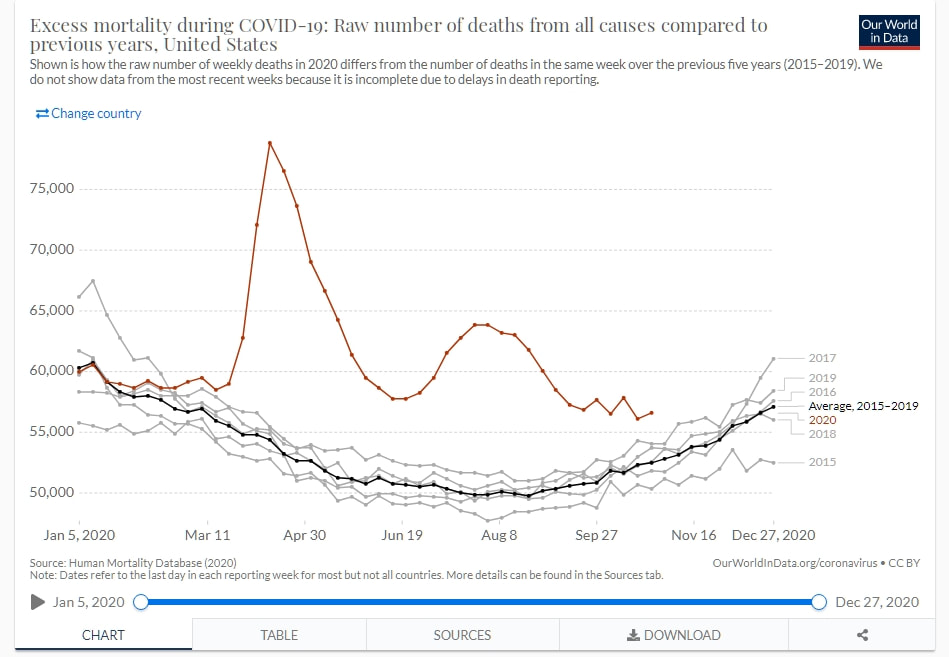
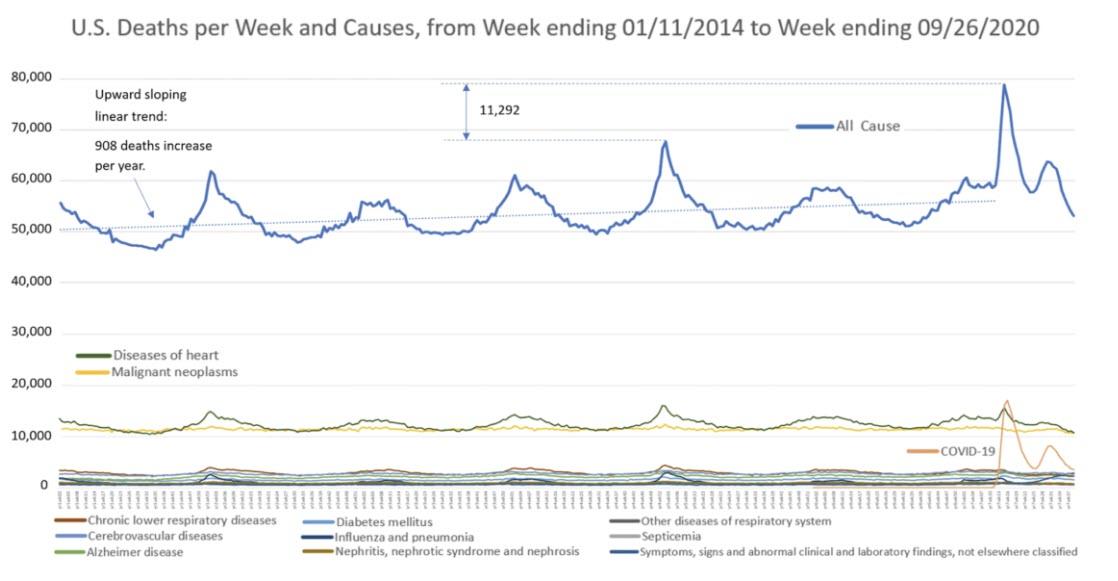
Ci sono oltre mille commenti per quest'articolo, e da questi ho rilevato alcuni dettagli interessanti: ovvero che la mortalità americana NON E' AFFATTO COSTANTE (vedi anche le figure sopra), per cui se c'é una scusa da fare, è quella di avere pubblicato da una supposta 'affidabile' professionista, un lavoro che non è affatto affidabile. Il lavoro originale infatti, come tradotto da Donchisciotte, diceva che letteralmente:
comedonchisciotte.org/un-analisi-approfondita-delle-morti-causate-dal-covid-19-negli-stati-uniti/
Briand ha anche evidenziato che i dati riportano fra le 50’000 e le 70’000 morti giornaliere, sia prima che dopo l’arrivo del COVID-19, ad indicare che questo numero di decessi era normale ben prima che si manifestasse il COVID-19. Quindi, secondo Briand, non solo il COVID-19 non ha avuto effetto sulla percentuale di decessi fra gli anziani, ma non ha neppure aumentato il numero totale delle morti.
“Stando a questi numeri non ci sono prove che il COVID-19 abbia provocato decessi aggiuntivi. Il totale delle morti [durante il periodo in esame] non supera il numero consueto. Non abbiamo trovato prove del contrario,” ha concluso Briand.
The News-Letter ha chiesto a Briand se i numeri relativi alle morti da COVID-19 possano essere ritenuti fuorvianti, considerando che l’infezione potrebbe aver aggravato patologie croniche pregresse, provocando così il decesso dei pazienti.
“Se [il numero totale di decessi da COVID-19] non fosse fuorviante, quello che avremmo dovuto riscontrare sarebbe stato un aumento contemporaneo dei decessi da infarto e da COVID-19. Ma il fatto che gli infarti siano calati, come anche le altre cause tradizionali di decesso, non ci lascia altra scelta che pensare ad un qualche tipo di classificazione errata,” ha risposto Briand.
In parole povere l’incidenza del COVID-19 sui decessi negli Stati Uniti, sarebbe da considerare problematica solo se generasse un aumento del tasso di mortalità, che andasse a sommarsi a quello derivante dalle normali cause di morte. Dato che il numero di decessi dovuti a tutte le possibili cause è rimasto lo stesso sia prima che dopo l’arrivo del COVID-19, è difficile dire, secondo Briand, che il numero di morti causate da COVID-19 sia preoccupante.
Nei commenti dell'articolo ci sono posizioni molto diversificate. Non in quello di Comedonchisciotte, dove sono quasi tutti a favore della linea editoriale del sito: forse perché se non sei negazionista non duri a lungo, chissà...
Da questi commenti si vede sopratutto la linea tra repubblicani e democratici. Ma al dunque, stando a quel che hanno visto al riguardo, la mortalità americana pare che stia in questi termini:
Matt Russ Littler • 4 days ago
Russ, I've gone through the CDC's numbers year by year back to the 1930's. Your argument falls apart for the USA because 2019 to 2020 is the highest rise in all-cause mortality from one year to the next in that entire time span. Most years fall within 2.5% of the previous year. The highest was 4.5%. This year will exceed 7%. There's a lot of Americans dying unfortunately. I haven't performed a detailed analysis of other countries, nor do I need to, because my main concern is what's happening in my home country.
Mike Russ Littler • 4 days ago
I looked at the ONS data. There is an excess of 62,638 deaths for the first 47 weeks of this year in England and Wales, compared to the average for those 47 weeks in the previous 5 years. That was until November 20th. On that day there were 54,831 deaths from COVID-19 confirmed officially, so if anything, I would have expected a smaller excess. The excess is huge, so not sure how you could say the deaths were lower.
mentalplex Starchild • 5 days ago
To respond directly, your suggestion is nonsensical: you are saying that because restrictions mandated by the Swedish state prevented a greater death toll, that is evidence that mandatory restrictions are not useful. Or, charitably, you may be saying that because Swedish restrictions made a big difference, that is evidence that further restrictions would not be beneficial. Neither of those arguments makes any sense. I'm not sure where to begin debunking them.
Generally, I'm not interested in going through an entire lockdown debate with you in a Disqus comment thread. You've changed the subject. You said the Imperial college projections of what would happen without mitigation were "wildly inaccurate", and then gave a logically flawed example of the outcome given mitigation. I posted to point out the obvious logical error.
If you would like to learn more about what restrictions have been in place, where, I will share some links with you.
Re: comparing restrictions between US jurisdictions and Sweden, data on school status is here: https://www.edweek.org/ew/s... Most states do not have an order to close in person secondary schools. Sweden ordered in person secondary schools and post secondary schools closed early in the pandemic.
You can see a description of various countries' general status here: https://www.northstarmeetin...
Currently, Sweden has limited gatherings to 8. Early in the pandemic they limited gatherings to 50. There has never been a US federal order to prevent gathering sizes (though there have been federal guidelines suggesting gatherings be limited). The current US president has, in fact, held regular events outside the Swedish restrictions throughout the pandemic.
Beyond that, you haven't addressed the initial logical error I brought to your attention, and have changed the subject. I'm not really interested in engaging further.
Matt Brent Tharp • 5 days agoFor the USA, the death rate in 1956 was 9.26 deaths per 1k, and in 1957 it was 9.49 per 1k. That's an increase of 2.5%. In 1966, the death rate was 9.48 per 1k, in 1967, 9.31 per 1k. A decline of 1.8%. In 2019, the death rate was 8.7 per 1k, and 2020 is on pace to have a death rate of 9.3 per 1k, an increase of 6.9%. That would be the largest rise from one year to the next since at least the 1930's. I haven't found data for earlier than that.
"Obviously, if it were as bad as 1957 and 1967, everyone would know someone"
I haven't known a single person who's died of the flu, in 38 years. I know three people who died from COVID-19 this year. Anecdotal evidence isn't the best evidence.
pgb Tom L • 3 days ago
Weekly data is downloadable from the CDC website. We are currently at 2,851,262 deaths through Nov 14th.
You can download the data and compare to 2019 and 2018. We are 342,235 deaths ahead of 2019 and 347,854 ahead of 2016. That's about a 12% increase.
In case you were wondering, the biggest increase we had ever had previously was 85k between 2014 and 2015.
TNUGA Maxwell • 8 days ago
"2020: as of 11/14 total deaths= 2,512,880"
You cite no source for that, but that number indicates Total Deaths since Feb 1, not since January 1. As of today, total deaths from all causes are 2,755,735 for Calendar 2020.
Total deaths in Fiscal 2020 (October 1 2019, to September 30, 2020 were 3,137,889. That's an increase of 306,656 over fiscal 2019!
Your premise is inaccurate!
Fiscal 2017: 2,790,291
Fiscal 2018: 2,835,736
Fiscal 2019: 2,831,233
Fiscal 2020: 3,137,889
Matt Maxwell • 7 days ago • editedI don't think your number for 2020 includes the first three weeks of January, or at least, the CDC's numbers for total deaths and COVID-19 starts with the final week of January.
https://www.cdc.gov/nchs/nv...
2,590,780 deaths as of Nov 28. 332nd day of the year, and missing the first three weeks of January, so that's total deaths in the last 308 days. 2590780 / 308 = 8411 deaths per day. There's also a lag time of 8 weeks before all death certificates are accounted for, so that number will rise. Also, January and December are typically the highest mortality months.
2020: 8411 deaths per day (for now)
2019: 7822 deaths per day
2018: 7778 deaths per day.
2017: 7708 deaths per day
2016: 7518 deaths per day
I'm not going to keep going, nor did I verify your numbers for the previous years, but as of right now, 2020 is up 7.5% from the previous year. Note the CDC actually lists excess deaths at 12%, and once January and December are taken into account, that may be accurate. I'm not sure why you're getting so many thumbs up for information that's clearly not accurate and no source is provided.
Edit: Now that I've read some of the other comments, you've been informed numerous times that your numbers are inaccurate. I think the correct thing to do would be to edit your post and provide the correct information.
AVG Age of "Death by Covid":
Austria 80+ years Source EMS;
Canada 86 years Source HCSC;
England 82 years Source NHS;
France 84 years Source SPF;
Germany 82 years Source RKI;
Italy 81 years Source ISS;
Spain 82 years Source MDS;
Switzerland 84 years Source BAG;
United States 80 years Source CDC;
Sweden 86 years Source FOHM
Matt Lord Rothschild, • 7 days ago • editedI went back to the 1930's. The largest increase in deaths from one year to the next was 4.5%. That's almost 100 years of data, and the absolute worst was 4.5%. 2019 to 2020 is on pace to exceed 7%. You're going to claim with a straight face that her quote is correct about a year that's going to be the worst in a century? Come on, man.
Adesso ci si potrà chiedere perché ci siano tante persone così poco convinte dalla scienza. Per esempio, perché fare un vaccino è visto da molti come un pericolo piuttosto che un risultato utile? Forse perché i no-vax hanno avuto molte ragioni in passato per essere scettici verso le campagne 'salutiste' dei governi, per esempio.
***********************************************************************************************************************************************************************************
Spesso si sente parlare di sfiducia nella scienza medica. Ma è una sfiducia immeritata? Alle volte è più che comprensibile.
Questo è un documentato studio di Harvard sulle campagne sanitarie fatte dai colonialisti europei sugli africani negli anni '20-'50. L'unica cosa che mi sento di aggiungere a questo orrore è che esistono effettivamente dei sospetti sull'espansione dell'HIV tramite campagne vaccinali, incluso un esperimento di un terzo vaccino antipolio, fatto proprio in Belgio. Ma questa è un'altra storia.
Colonial Medical Campaigns
French, British, and Belgian colonial governments implemented a wide variety of medical campaigns beginning in the early 20th century.8 The introduction of these efforts coincided with greater European penetration into rural areas and to large outbreaks of human African trypanosomiasis, also known as sleeping sickness. The largest and most pervasive medical campaigns organized by the French focused on the treatment and prevention of sleeping sickness. However, the campaigns also targeted other diseases including yaws, malaria, leprosy, and yellow fever
(Headrick, 1994, 2014; Pépin, 2011).
Sleeping sickness is a lethal parasitic disease transmitted by the bite of a tsetse fly, which is only present in Africa. There are two stages of the disease. An individual in the first stage 8For an overview of the approaches used by various colonizing countries, see Headrick (2014). In short, while the French focused on treating the individual, the British focused on controlling the disease environment. The Belgians largely modeled their health initiatives after the French of the disease experiences joint pain, headaches, and fever. The disease can cause drowsiness and swelling in the lymph nodes. In the second stage, the disease infects the nervous system, and the individual experiences extreme lethargy and eventually dies. There are two types of human sleeping sickness. The more acute and rapid acting form of the disease, Trypanosoma brucei rhodesiense, is found in Eastern and Southern Africa. However, most sleeping sickness cases in humans are from the chronic form of the disease, Trypanosoma brucei gambiense, which is found in Western and Central Africa. There is also a form of sleeping sickness that affects domesticated animals, Trypanosoma brucei brucei, which is also known as nagana. The sleeping sickness epidemics motivated a large European response during the colonial era. This was partially due to humanitarian concerns but also due to concerns about labor supply, particularlyin the sparsely populated equatorial zone. Scientific and nationalistic motivations were also important, as the colonial governments competed over developing advances in tropical medicine
(Headrick, 1994, 2014).
In French colonies, the military organized and implemented campaigns through a system of mobile medical teams that focused exclusively on sleeping sickness. In Cameroon, the mobile medical teams were first organized in 1921. AEF organized mobile teams, called the Service de la prophylaxie de la trypanosomiase (Trypanosomiasis prophylaxis service), beginning in 1927. The mobile teams generally consisted of one French military doctor, several African nurses, two white corporals, several African soldiers and a large number of porters to carry equipment. The teams faced the challenging task of visiting rural villages at a time of minimal road infrastructure.
During a medical team’s visit to a village, villagers were forced, often at gunpoint, to undergo a physical examination. The examinations included neck palpitations to check for swelling of the lymph nodes, blood tests to check for trypanosomes in the blood, and spinal taps. If an individual
was believed to have sleeping sickness, an effort was made to determine whether the patient was in the first or second stage of the disease. However, this diagnosis process was often imperfect (Headrick, 1994, 2014).
The campaigns initially focused exclusively on the treatment of sleeping sickness. One of the earliest forms of treatment for sleeping sickness was the drug atoxyl, an arsenic based drug.
While the name atoxyl literally means non-toxic, the drug had a chemotherapeutic index close to one. This means that the dose of treatment required to rid the body of the trypanosomes was almost equal to the dose that would be lethal to the patient. Additionally, the drug caused partial or total blindness in up to 20% of patients (Headrick, 2014). The drug was administered to patients regardless of whether they were known to have sleeping sickness. While the drug was effective in treating the disease during the first stage, it could actually increase speed of death in the second stage. However, accurate staging of the disease was difficult, particularly in the field. The coverage of the campaigns was impressive. For example, in Cameroon in 1928, the mobile medical teams examined 663,971 people, of whom 17% were identified as having sleeping sickness (Le Gouvernement Fraçais, 1929).
Despite the negative side effects of atoxyl, the French continued to use the drug well into the 1930’s, while in non-French colonies it had stopped being used a decade earlier (Headrick, 1994,p. 320). There were several key factors driving the use of atoxyl. First, atoxyl was very cheap relative to any alternatives, almost seven times cheaper than the next cheapest drug. Second, it was relatively easy to use. This was a particularly important consideration given that the mobile teams worked in difficult environments and that local nurses were often administering the shots. For example, tryparsamide, a less toxic alternative to atoxyl, could cause immediate skin reactions in patients, leading to abscessing at the injection site if the needle and skin were not perfectly clean. Finally, atoxyl was relatively, though not completely, stable in tropical environments. As described by Headrick (1994, p. 330): "Atoxyl’s grip remained because its price and convenience were unbeatable. The health service tolerated accidents and blindness because they did not usually appear at the moment of injection, unlike an abscess from a faulty tryparsamide shot.” More broadly, the campaigns themselves were less concerned with the health of individual patients, than the broader public health effects of the campaigns. The doctors in charge of the campaigns were willing to tolerate the negative effects on individual patients if it meant that other individuals were then less likely to get sleeping sickness.
Subsequent medications for sleeping sickness, such as Lomidine (also known as Pentamidine in the United States), were less toxic, but often had serious side effects. Lomidine was believed to work as a prophylactic, which means it was supposed to prevent individuals from getting sleeping sickness, rather than treating those who already had sleeping sickness. During the campaigns, all individuals in a village were required to receive Lomidine injections. The Lomidine injection needed to be administered every six months in order for it to effectively prevent sleeping sickness in an individual. Even though Lomidine was believed to prevent the spread of sleeping sickness, it was also associated with significant side effects. The injections themselves were painful and caused dizziness and low blood pressure. Entire villages were required to rest under the supervision of the medical team after receiving the injections. Lomidine injections were also associated with several serious accidents, including the development of gangrene at the injection site and death. In fact, Lomidine was considered too dangerous for use on Europeans. Ultimately, Lomidine was shown to be ineffective at prevention, but for a short term would reduce the number of trypanosomes circulating in the blood(Lachenal, 2017, p.174). In fact, in 1974, a French doctor involved with the colonial medical campaigns declared that the Lomidine injections were ”pointless, dangerous, and therefore pointlessly dangerous” (Lachenal, 2017, p.182).
Historians and anthropologists have linked the sleeping sickness campaigns to mistrust in modern medicine, as individuals were often forced to participate in the campaigns and the treatments had severe negative side effects. Furthermore, the efficacy of the drugs used in the
campaigns was dubious. Anecdotally, the experience of these campaigns has affected present day views of medicine. Feldman-Savelsberg et al. (2000, p. 162) explain resistance to a tetanus
In 2018, results were released from a medical trial that suggest that a new orally administered drug, fexinidazole, can effectively treat late stage sleeping sickness, a breakthrough relative to the present first line treatment therapy of oral nifurtimox and intravenous eflornithine that must be administered in a hospital setting (Mesu et al., 2018;Maxmen, 2017).10
campaign in Cameroon in 1990 by noting that "[the modern medical campaigns]...awakened negative collective memories of French colonial efforts to wipe out sleeping sickness". Similarly, Giles-Vernick (2002, p. 106) reports on rumors and memories that were still circulating in the late 1980s in the Nola region of Central African Republic (CAR) that the injections for sleeping sickness brought death. In fact, the Eton ethnic group from Central Cameroon has a song about
the sleeping sickness campaigns and the negative side effects of the sleeping sickness injections.
Part of the song lyrics are as follows:
The injection against sleeping sickness was too painful
The injection against sleeping sickness was too painful
They gave me an injection in the head
They gave me an injection in the neck
They gave me an injection in the back
...
They ask me to go draw water from the well
If I drag my feet
The policemen hit me on the head
The injection against sleeping sickness was too painful (Lachenal, 2014)
The song highlights that the memory of the sleeping sickness campaigns remain, how memories of the campaigns may be transmitted across generations, and that the campaigns were characterized as painful by the participants. Additionally, epidemiologists have examined the effects of the unsanitary practices used during the campaigns on the spread of contagious disease. While the campaigns followed standard contemporaneous medical procedures, they may have contributed to the proliferation of certain blood-born diseases from the reuse of unsanitary needles (Pépin, 2011). For example, campaigns against schistosomiasis in Egypt have been associated with the iatrogenic spread (illness related to medical practice or treatment) of Hepatitis C (Frank et al., 2000). Medical researchers have documented a link between exposure to colonial medical campaigns and Hepatitis C virus (HCV) infection rates in Cameroon, which today has one of the highest Hepatitis C infection rates in the world (Nerrienet et al., 2005).10 Pépin and Labbé (2008) and Pépin et al. (2010) link HCV and Human T-Cell Lymphotropic Virus (a retrovirus that causes adult T-cell leukemia, henceforth HTCLV) rates in former AEF countries and Cameroon to colonial medical campaign exposure. Additionally, Pépin (2011) hypothesizes that in AEF, the medical campaigns may have contributed 10Epidemiologists often use HCV to examine iatrogenic transmissions of diseases because HCV is generally nonlethal and difficult to spread through sex. to the initial spread of HIV prior to its initial identification, as it gave the virus access to large swaths of population to which it would not have otherwise had access.
scholar.harvard.edu/files/slowes/files/lowes_montero_colonialmedicine.pdf
**************************************************************************************************************************************************************************************
MA ci si poteva aspettare che i test sui vaccini fossero messi in discussione pure sul British Medical Journal? Forse forse Cristanti non aveva tutti i torti sull'affidabilità dei numeri dei vaccini?
Will covid-19 vaccines save lives? Current trials aren’t designed to tell us
BMJ 2020; 371 doi: https://doi.org/10.1136/bmj.m4037 (Published 21 October 2020)
The world has bet the farm on vaccines as the solution to the pandemic, but the trials are not focused on answering the questions many might assume they are. Peter Doshi reports
As phase III trials of covid-19 vaccines reach their target enrolments, officials have been trying to project calm. The US coronavirus czar Anthony Fauci and the Food and Drug Administration leadership have offered public assurances that established procedures will be followed.1234 Only a “safe and effective” vaccine will be approved, they say, and nine vaccine manufacturers issued a rare joint statement pledging not to prematurely seek regulatory review.5
But what will it mean exactly when a vaccine is declared “effective”? To the public this seems fairly obvious. “The primary goal of a covid-19 vaccine is to keep people from getting very sick and dying,” a National Public Radio broadcast said bluntly.6
Peter Hotez, dean of the National School of Tropical Medicine at Baylor College of Medicine in Houston, said, “Ideally, you want an antiviral vaccine to do two things . . . first, reduce the likelihood you will get severely ill and go to the hospital, and two, prevent infection and therefore interrupt disease transmission.”7
Yet the current phase III trials are not actually set up to prove either (table 1). None of the trials currently under way are designed to detect a reduction in any serious outcome such as hospital admissions, use of intensive care, or deaths. Nor are the vaccines being studied to determine whether they can interrupt transmission of the virus.
Evaluating mild, not severe, disease
In a September interview Medscape editor in chief Eric Topol pondered what counts as a recorded “event” in the vaccine trials. “We’re not talking about just a PCR [polymerase chain reaction test]-positive mild infection. It has to be moderate to severe illness to qualify as an event, correct?” he asked.8
“That’s right,” concurred his guest, Paul Offit, a vaccinologist who sits on the FDA advisory committee that may ultimately recommend the vaccines for licence or emergency use authorisation.
But that’s not right. In all the ongoing phase III trials for which details have been released, laboratory confirmed infections even with only mild symptoms qualify as meeting the primary endpoint definition.9101112 In Pfizer and Moderna’s trials, for example, people with only a cough and positive laboratory test would bring those trials one event closer to their completion. (If AstraZeneca’s ongoing UK trial is designed similarly to its “paused” US trial for which the company has released details, a cough and fever with positive PCR test would suffice.)
Part of the reason may be numbers. Severe illness requiring hospital admission, which happens in only a small fraction of symptomatic covid-19 cases, would be unlikely to occur in significant numbers in trials. Data published by the US Centers for Disease Control and Prevention in late April reported a symptomatic case hospitalisation ratio of 3.4% overall, varying from 1.7% in 0-49 year olds and 4.5% in 50-64 year olds to 7.4% in those 65 and over.13 Because most people with symptomatic covid-19 experience only mild symptoms,14 even trials involving 30 000 or more patients would turn up relatively few cases of severe disease.
In the trials, final efficacy analyses are planned after just 150 to 160 “events,”—that is, a positive indication of symptomatic covid-19, regardless of severity of the illness.
Yet until vaccine manufacturers began to release their study protocols in mid-September, trial registries and other publicly released information did little to dispel the notion that it was severe covid-19 that the trials were assessing. Moderna, for example, called hospital admissions a “key secondary endpoint” in statements to the media.15 And a press release from the US National Institutes of Health reinforced this impression, stating that Moderna’s trial “aims to study whether the vaccine can prevent severe covid-19” and “seeks to answer if the vaccine can prevent death caused by covid-19.”16
But Tal Zaks, chief medical officer at Moderna, told The BMJ that the company’s trial lacks adequate statistical power to assess those outcomes. “The trial is precluded from judging [hospital admissions], based on what is a reasonable size and duration to serve the public good here,” he said.
Hospital admissions and deaths from covid-19 are simply too uncommon in the population being studied for an effective vaccine to demonstrate statistically significant differences in a trial of 30 000 people. The same is true of its ability to save lives or prevent transmission: the trials are not designed to find out.
Zaks said, “Would I like to know that this prevents mortality? Sure, because I believe it does. I just don’t think it’s feasible within the timeframe [of the trial]—too many would die waiting for the results before we ever knew that.”
Stopping transmission
What about Hotez’s second criterion, interrupting virus transmission, which some experts have argued17 should be the most important test in phase III studies?
“Our trial will not demonstrate prevention of transmission,” Zaks said, “because in order to do that you have to swab people twice a week for very long periods, and that becomes operationally untenable.”
He repeatedly emphasised these “operational realities” of running a vaccine trial. “Every trial design, especially phase III, is always a balancing act between different needs,” he said. “If you wanted to have an answer on an endpoint that happens at a frequency of one 10th or one fifth the frequency of the primary endpoint, you would need a trial that is either 5 or 10 times larger or you’d need a trial that is 5 or 10 times longer to collect those events. Neither of these, I think, are acceptable in the current public need for knowing expeditiously that a vaccine works.”
Zaks added, “A 30 000 [participant] trial is already a fairly large trial. If you’re asking for a 300 000 trial then you need to talk to the people who are paying for it, because now you’re talking about not a $500m to $1bn trial, you’re talking about something 10 times the size. And I think the public purse and operational capabilities and capacities we have are rightly spent not betting the farm on one vaccine but, as Operation Warp Speed [the US government’s covid-19 vaccine plan] is trying to do, making sure that we’re funding several vaccines in parallel.”
Debating endpoints
Still, it’s fair to say that most of the general public assumes that the whole point of the current trials, besides testing safety (box 1), is to see whether the vaccine can prevent bad outcomes. “How do you reconcile that?” The BMJ asked Zaks.
History shows many examples of serious adverse events from vaccines brought to market in periods of enormous pressure and expectation. There were contaminated polio vaccines in 1955, cases of Guillain-Barré syndrome in recipients of flu vaccines in 1976, and narcolepsy linked to one brand of influenza vaccine in 2009.
“Finding severe rare adverse events will require the study of tens of thousands of patients, but this requirement will not be met by early adoption of a product that has not completed its full trial evaluation,” Harvard drug policy researchers Jerry Avorn and Aaron Kesselheim recently wrote in JAMA.20
Covid-19 vaccine trials are currently designed to tabulate final efficacy results once 150 to 160 trial participants develop symptomatic covid-19—and most trials have specified at least one interim analysis allowing for the trials to end with even fewer data accrued.
Medscape’s Eric Topol has been a vocal critic of the trials’ many interim analyses. “These numbers seem totally out of line with what would be considered stopping rules,” he says. “I mean, you’re talking about giving a vaccine with any of these programmes to tens of millions of people. And you’re going to base that on 100 events?”
Great uncertainty remains over how long a randomised trial of a vaccine will be allowed to proceed. If efficacy is declared, one possibility is that the thousands of volunteers who received a saline placebo would be offered the active vaccine, in effect ending the period of randomised follow-up. Such a move would have far reaching implications for our understanding of vaccines’ benefits and harms, rendering uncertain our knowledge of whether the vaccines can reduce the risk of serious covid-19 disease and precluding any further ability to compare adverse events in the experimental versus the placebo arm.
“It’ll be a decision we’ll have to take at that time. We have not committed one way or another,” Moderna’s Tal Zaks told The BMJ. “It will be a decision where FDA and NIH will also weigh in. And it will be probably a very difficult decision, because you will be weighing the benefit to the public in continuing to understand the longer term safety by keeping people on placebo and the expectation of the people who have received placebo to be crossed over now that it has been proved effective.”
“Very simply,” he replied. “Number one, we have a bad outcome as our endpoint. It’s covid-19 disease.” Moderna, like Pfizer and Janssen, has designed its study to detect a relative risk reduction of at least 30% in participants developing laboratory confirmed covid-19, consistent with FDA and international guidance.2122
Number two, Zaks pointed to influenza vaccines, saying they protect against severe disease better than mild disease. To Moderna, it’s the same for covid-19: if its vaccine is shown to reduce symptomatic covid-19, it will be confident it also protects against serious outcomes.
But the truth is that the science remains far from clear cut, even for influenza vaccines that have been used for decades. Although randomised trials have shown an effect in reducing the risk of symptomatic influenza, such trials have never been conducted in elderly people living in the community to see whether they save lives.
Only two placebo controlled trials in this population have ever been conducted, and neither was designed to detect any difference in hospital admissions or deaths.23 Moreover, dramatic increases in use of influenza vaccines has not been associated with a decline in mortality (box 2).26
Not enrolling enough elderly people or minorities
A vaccine that has been proved to reduce the risk of symptomatic disease by a certain proportion should, you might think, reduce serious outcomes such as hospital admissions and deaths in equal proportion.
Peter Marks, an FDA official with responsibility over vaccine approvals, recently stated as much about influenza vaccination, which “only prevents flu in about half the people who get it. And yet that’s very important because that means that it leads to half as many deaths related to influenza each year.”
But when vaccines are not equally effective in all populations the theory breaks down.
If frail elderly people, who are understood to die in disproportionate numbers from both influenza and covid-19, are not enrolled into vaccine trials in sufficient numbers to determine whether case numbers are reduced in this group, there can be little basis for assuming any benefit in terms of hospital admissions or mortality. Whatever reduction in cases is seen in the overall study population (most of which may be among healthy adults), this benefit may not apply to the frail elderly subpopulation, and few lives may be saved.
This is hard to evaluate in the current trials because there are large gaps in the types of people being enrolled in the phase III trials (table 1). Despite recruiting tens of thousands, only two trials are enrolling children less than 18 years old. All exclude immunocompromised people and pregnant or breastfeeding women, and though the trials are enrolling elderly people, few or perhaps none of the studies would seem to be designed to conclusively answer whether there is a benefit in this population, despite their obvious vulnerability to covid-19.
“Adults over 65 will be an important subgroup that we will be looking at,” Moderna’s Zaks told The BMJ. “That said . . . any given study is powered for its primary endpoint—in our case covid-19 disease irrespective of age.”
Al Sommer, dean emeritus of the Johns Hopkins School of Public Health, told The BMJ, “If they have not powered for evidence of benefit in the elderly, I would find that a significant, unfortunate shortcoming.” He emphasised the need for “innovative follow-up studies that will enable us to better determine the direct level of protection immunisation has on the young and, separately, the elderly, in addition to those at the highest risk of severe disease and hospitalisation.”
One view is that trial data should be there for all target populations. “If we don’t have adequate data in the greater than 65 year old group, then the greater than 65 year old person shouldn’t get this vaccine, which would be a shame because they’re the ones who are most likely to die from this infection,” said vaccinologist Paul Offit.8 “We have to generate those data,” he said. “I can’t see how anybody—the Data and Safety Monitoring Board or the FDA Vaccine Advisory Committee, or FDA decision-makers—would ever allow a vaccine to be recommended for that group without having adequate data.”
“I feel the same way about minorities,” Offit added. “You can’t convince minority populations to get this vaccine unless they are represented in these trials. Otherwise, they’re going to feel like they’re guinea pigs, and understandably so.”
**************************************************************************************************************************************************************************************
MA poi tutti i morti dell'era COVID... sono solo di COVID? Ovviamente no. Più la pandemia va avanti... e più soffrono anche le altre patologie. E' incerto se queste morti siano da attribuire al COVID come indirette oppure no, ma restano sempre morti che si sarebbero dovute evitare. Negli USA già a settembre c'erano 273.000 morti in più del normale, ma soltanto 220k circa erano dovuti al COVID. E gli altri?
www.washingtonpost.com/graphics/2020/investigations/coronavirus-excess-deaths-heart/#comments-wrapper
Heart conditions drove spike in deaths beyond those attributed to covid-19, analysis shows
Fear of seeking care in hospitals overwhelmed by the
pandemic may have caused thousands of deaths, experts say
Image: Joseph Neufeld, Jr. looks over caskets at the Gerard J. Neufeld funeral home in the New York borough of Queens in late April.
Joseph Neufeld, Jr. looks over caskets at the Gerard J. Neufeld funeral home in the New York borough of Queens in late April. (Bryan R Smith/Reuters)
By Reis Thebault, Lenny Bernstein, Andrew Ba Tran and Youjin Shin
July 2, 2020
Home
Share
545
The coronavirus killed tens of thousands in the United States during the pandemic’s first months, but it also left a lesser-known toll: thousands more deaths than would have been expected from heart disease and a handful of other medical conditions, according to an analysis of federal data by The Washington Post.
The analysis suggests that in five hard-hit states and New York City there were 8,300 more deaths from heart problems than would have been typical in March, April and May — an increase of roughly 27 percent over historical averages.
[Stay safe and informed with our free Coronavirus Updates newsletter]
That spike contributed to Illinois, Massachusetts, Michigan, New Jersey, and New York state and the city having a combined 75,000 “excess deaths” during that period, 17,000 more than the number officially attributed to covid-19, the disease the virus causes.
While several experts said some of the excess deaths in the analysis were almost certainly unrecognized fatalities from covid-19, the review suggests that many patients suffering from serious conditions died as a result of delaying or not seeking care as the outbreak progressed and swamped some hospitals.
Normally, heart disease is the leading cause of death in the United States. But in the early months of the pandemic, some hospital departments were nearly devoid of the heart, cancer, stroke and other patients who populated them before.
Looking at the analysis, more than 50 patients a day “died excess deaths just from heart disease, just in New York City,” said John Puskas, chairman of cardiovascular surgery at Mount Sinai Morningside Hospital in Manhattan. “Frankly, that would explain where all the patients went.”
The analysis of data from March 1 to May 30, using a model previously developed by the Yale School of Public Health, shows heart disease is the major driver of excess deaths, excluding those officially attributed to covid-19.
As states in the South and West restart their economies and see new cases surge, the excess deaths should serve as a cautionary tale, said Nahid Bhadelia, medical director of Boston University School of Medicine’s Special Pathogens Unit.
“This data underlines the importance of not letting our health systems get to the point where they are so overwhelmed that it spills over and affects people with other medical conditions in our community,” she said.
The number of excess deaths in the United States during the pandemic far surpasses the number officially attributed to covid-19, analyses have shown. The official death counts from the disease are incomplete, according to experts, in part because it spread for weeks before testing was widely available and because the virus kills in a variety of ways that were not recognized early on.
But several experts said the spikes in the causes of death in the new Post analysis suggested a deadly collateral effect of the pandemic. They said the surge in deaths from heart disease and several other conditions matches what they saw in clinics and hospitals and confirms their fears that many patients died after not seeking care.
“This is in line with what we were scared of happening: that we were missing people beforehand and that people were dying of other diseases,” Bhadelia said.
New York City, which reports its covid-19 statistics separately from the state, quickly became the epicenter of the pandemic in the United States and saw more than 4,700 excess deaths from heart disease — more than four times the number of any other jurisdiction The Post examined.
Puskas said that even at the height of the outbreak, when his hospital was nearly overwhelmed by the pandemic response, it didn’t turn away anyone seeking heart care. Yet the number of cardiovascular patients showing up remained low, he said.
Health-care providers everywhere are now reckoning with the consequences.
“All those patients that would typically have been there having cardiovascular care were not there,” Puskas said. “Those who would’ve had emergency lifesaving care did not receive that care, and they then became one of the statistics on your chart.”
In many cases, experts said, patients suffered through cardiac events, strokes, hyperglycemia and other health difficulties at home, likely fearful of seeking care in hospitals where large numbers of people suffering from covid-19 were receiving treatment.
Between March 15 and May 23, visits to hospital emergency departments declined 23 percent for heart attacks, 20 percent for strokes and 10 percent for high-blood-sugar crises — a complication of diabetes — when compared with the previous 10 weeks, according to a report last month from the Centers for Disease Control and Prevention. “The striking decline in ED visits for acute life-threatening conditions might partially explain observed excess mortality not associated with COVID-19,” the researchers wrote.
And in a separate excess deaths analysis, the CDC estimated that since Feb. 1, between about 20,000 and 49,000 more people have died of all non-covid-19 causes than would be expected in a typical year.
On Wednesday, an article published in the Journal of the American Medical Association bolstered the findings from the Post and CDC analyses. The paper looked at excess deaths nationwide in March and April and found that 35 percent were attributed to causes other than covid-19. The researchers, led by a team at Virginia Commonwealth University, concluded that those deaths may include unreported, “nonrespiratory manifestations” of the virus, or they could represent “secondary pandemic mortality caused by disruptions in society that diminished or delayed access to health care.”
The emerging understanding of the collateral toll signals the fallout that America’s new hot spots could face in coming weeks and also suggests the backlog in health-care problems that hospitals and other caregivers will face once the pandemic subsides.
“We are waiting for this cloud of coronavirus to lift more fully so we can see what’s happened to our patients,” said Gregory Piazza, a cardiovascular specialist at Brigham and Women’s Hospital in Boston. “Are we going to see a flood of new patients that have decreased left heart function because they’ve had a missed myocardial infarction? Are we going to see more patients with heart failure?
“We’re certainly preparing, in the cardiovascular community, to run double speed to accommodate all the things we’ve put off, that have been backing up all this time,” he said.
The head of the National Cancer Institute, Norman “Ned” Sharpless, predicted on June 18 that delays in cancer screenings, diagnoses and treatment during the pandemic are likely to result in “thousands” of excess cancer deaths in years to come.
For its analysis, The Post selected the five states and New York City because their official covid-19 tolls ranked among the highest in the nation and their death data were among the most complete. The analysis focused on common causes of death.
The analysis calculates excess deaths in the United States by estimating the number of people who would have died absent the pandemic — an estimate that takes into account population growth and seasonal variations — and subtracting that number from the overall number of deaths reported by the National Center for Health Statistics (NCHS).
The great majority of those excess deaths are counted as covid-19 deaths. In the United States to date, at least 125,000 deaths are officially attributed to the disease, according to a Post tally.
Some of the remaining excess deaths — especially early in the pandemic — were likely caused by covid-19 but not attributed to the disease when death certificates were filled out by physicians, coroners or medical examiners.
“Not everyone gets an autopsy,” said Evert Eriksson, trauma medical director at the Medical University of South Carolina, one of the physicians who saw empty beds as the pandemic first hit. “There is some miscoding that goes on in there. That’s why a lot of this is difficult to tie causation to.”
Increases in deaths from pneumonia and influenza, for example, point clearly to missed cases of covid-19, said Dan Weinberger, professor of epidemiology at the Yale School of Public Health, who led the team that developed the model The Post used in its analysis and co-wrote the JAMA paper.
The Post found about 2,100 excess deaths from influenza and pneumonia in the six locations it examined, about 20 percent of them in New York City alone.
Because tracking efforts showed that influenza fell to “undetectable levels” in March, Weinberger said, “the excess pneumonia and influenza deaths we’re seeing are probably covid-19 deaths that are just not being attributed to the virus.”
Variations in the way underlying causes of death are recorded on some death certificates mean some covid-19 deaths could be included in the analysis of other causes; however, the number is small and does not account for the dramatic increase in excess deaths from heart disease and other causes, said Robert N. Anderson, chief of the Mortality Statistics Branch of the National Center for Health Statistics.
Piazza said he was not surprised to see across-the-board increases in diabetes deaths given that the pandemic has drastically altered routines, making it more difficult for people to stick to regular diets and exercise crucial for glucose control. Some diabetics may be reluctant to go to the pharmacy and could be more likely to run out of medication, he said.
The Post analysis estimated there were more than 1,300 excess deaths from diabetes.
“Diabetes is the kind of illness that requires, for good control, a reasonable amount of monitoring from both patient and provider,” Piazza said.
The Post analysis also estimated there were 1,300 more deaths attributed to Alzheimer’s disease than would have been expected, adding to the pandemic’s devastating toll on the country’s elderly.
The coronavirus has made nursing homes some of the deadliest places, and responding to the crisis probably affected care across the board, Bhadelia said. Facilities may also have been reluctant to send uninfected residents to hospitals for fear of exposure, she said.
“They just didn’t get timely care,” Bhadelia said.
And as with other diseases, experts said, some of those Alzheimer’s deaths may have been missed cases of covid-19.
The Post’s analysis found that cerebrovascular diseases — stroke and other conditions that involve blood flow to the brain — accounted for more than 170 excess deaths in New York City and more than 120 in New Jersey.
In New York state and Illinois, the numbers were smaller, well below 100 each. Massachusetts and Michigan saw about the same or fewer deaths from strokes than would have been expected over the same period.
Though covid-19 was originally believed to be primarily a respiratory illness, Eriksson noted its ability to attack blood vessels and form blood clots. That virus-related complication could have created stroke symptoms in some patients, he said, but there also almost certainly were patients who delayed stroke treatment out of fear of exposure to the virus.
Analyzing deaths by cause underscores the severity of the pandemic, its cascading effects and the deadliness of the novel coronavirus, Weinberger said.
“It’s useful to see the magnitude of those increases in relation to the magnitude of the epidemic overall,” he said. “It tells us even if there are deaths due to these other causes, it’s probably much smaller than the deaths we’re seeing due to the virus.”
Lenny Bronner contributed to this report.
*************************************************************************************************************************************************************************************
MA le lezioni dal passato non finiscono certo qui. Quando c'é qualcuno che racconta storie su come sia impossibile che il SARS-COV-2 (AKA COVID-19) sia un germe sintetico dovrebbero a questo punto ricordarsi del perché questo possa essere possibile. Infatti, per chi non lo sapesse, sono riusciti a ricostruire addirittura il virus dell'influenza spagnola, sì... quello del 1918!
Come? Ce lo racconta direttamente il CDC:
www.cdc.gov/flu/pandemic-resources/reconstruction-1918-virus.html?fbclid=IwAR0fIlfHSREdk10WXNKmxJja7LxConBUVxby11DnHMptJ8Jtq0brbprA_Qo#reconstruction
The Deadliest Flu: The Complete Story of the Discovery and Reconstruction of the 1918 Pandemic VirusBy Douglas Jordan with contributions from Dr. Terrence Tumpey and Barbara Jester
On This Page
- Background: The deadly legacy of the 1918 pandemic and its importance for global efforts to prepare against future pandemic threats.
- Part 1 – Discovering a Lost Killer: The story of a virus hunter’s lifelong pursuit to discover the deadliest pandemic flu virus in human history.
- Part 2 – Building the Blueprint: The story of how a team of U.S. scientists decoded and assembled the genome of the 1918 virus.
- Part 3 – The Reconstruction: The story of how a CDC microbiologist reconstructed the live 1918 pandemic virus in a secure CDC laboratory to unravel its secrets and protect against future pandemics.
- Part 4 – Learning from the Past: How the world has progressed since the 1918 pandemic and the challenges posed by future pandemics.
- References
The 1918 virus caused the deadliest flu pandemic in recorded human history, claiming the lives of an estimated 50 million people worldwide.
resize iconView LargerThe 100-year anniversary of the 1918 pandemic and the 10-year anniversary of the 2009 H1N1 pandemic are milestones that provide an opportunity to reflect on the groundbreaking work that led to the discovery, sequencing and reconstruction of the 1918 pandemic flu virus. This collaborative effort advanced understanding of the deadliest flu pandemic in modern history and has helped the global public health community prepare for contemporary pandemics, such as 2009 H1N1, as well as future pandemic threats.
The 1918 H1N1 flu pandemic, sometimes referred to as the “Spanish flu,” killed an estimated 50 million people worldwide, including an estimated 675,000 people in the United States.1,2,3,4 An unusual characteristic of this virus was the high death rate it caused among healthy adults 15 to 34 years of age.3 The pandemic lowered the average life expectancy in the United States by more than 12 years.3 A comparable death rate has not been observed during any of the known flu seasons or pandemics that have occurred either prior to or following the 1918 pandemic.3
The virus’ unique severity puzzled researchers for decades, and prompted several questions, such as “Why was the 1918 virus so deadly?”, “Where did the virus originate from?”, and “What can the public health community learn from the 1918 virus to better prepare for and defend against future pandemics?” These questions drove an expert group of researchers and virus hunters to search for the lost 1918 virus, sequence its genome, recreate the virus in a highly safe and regulated laboratory setting at CDC, and ultimately study its secrets to better prepare for future pandemics. The following is a historical recounting of these efforts, complete with references and descriptions of the contributions made by all of the remarkable men and women involved.
Discovering a lost killer
Note: For a list of common questions and answers related to this work, see Q&A: Reconstruction of the 1918 Influenza Pandemic Virus.
This 1951 photo shows Johan Hultin (on left) and fellow university colleagues during his initial attempt to obtain the 1918 virus from bodies of victims buried in permafrost at the Brevig Mission burial site. Photo credit: Johan Hultin
resize iconView LargerFor decades, the 1918 virus was lost to history, a relic of a time when the understanding of infectious pathogens and the tools to study them were still in their infancy. Following the 1918 pandemic, generations of scientists and public health experts were left with only the epidemiological evidence of the 1918 pandemic virus’ lethality and the deleterious impact it had on global populations. A small ocean-side village in Alaska called Brevig Mission would become both testament to this deadly legacy as well as crucial to the 1918 virus’ eventual discovery.
Today, fewer than 400 people live in Brevig Mission, but in the fall of 1918, around 80 adults lived there, mostly Inuit Natives. While different narratives exist as to how the 1918 virus came to reach the small village – whether by traders from a nearby city who traveled via dog-pulled sleds or even by a local mail delivery person – its impact on the village’s population is well documented. During the five-day period from November 15-20, 1918, the 1918 pandemic claimed the lives of 72 of the villages’ 80 adult inhabitants.
Later, at the order of the local government, a mass grave site marked only by small white crosses was created on a hill beside the village – a grim monument to a community all but erased from existence. The grave was frozen in permafrost and left untouched until 1951. That year, Johan Hultin, a 25-year-old Swedish microbiologist and Ph.D. student at the University of Iowa, set out on an expedition to Brevig Mission in the hopes of finding the 1918 virus and in the process unearth new insights and answers. Hultin believed that within that preserved burial ground he might still find traces of the 1918 virus itself, frozen in time within the tissues of the villagers whose lives it had claimed.
In 1951, Hultin successfully obtained permission from the village elders to excavate the Brevig Mission burial site. With the help of several of his university colleagues, Hultin set up a dig site over the grave. The excavation took days, as Hultin had to create campfires to thaw the earth enough to allow for digging. Two days in, Hultin came across the body of a little girl — her body was still preserved wearing a blue dress, and her hair was adorned with red ribbons5. Ultimately, Hultin successfully obtained lung tissue from four additional bodies buried at the site, but logistical and technological limitations of the time period would prove formidable.
In a conversation Hultin had decades later with CDC microbiologist Dr. Terrence Tumpey (see part III – the reconstruction), Hultin would explain how during the return trip from Alaska to the University of Iowa, he flew on a DC-3 propeller-driven airplane that was forced to make multiple stops along the trip to refuel. During each stop, Hultin – ever resourceful – would deboard the plane and attempt to re-freeze the lung samples using carbon dioxide from a fire extinguisher.
The noise generated from this activity apparently drew puzzled glances from fellow passengers and onlookers. Once back in Iowa, Hultin attempted to inject the lung tissue into chicken eggs to get the virus to grow.5 It did not. In the end, perhaps unsurprisingly, Hultin was unable to retrieve the 1918 virus from this initial attempt.
A picture of Johan Hultin working in the laboratory in 1951. Hultin’s initial attempt to rescue the 1918 virus was unsuccessful. Note: using one’s mouth to draw virus into a pipette is not considered a safe laboratory practice today. Laboratory safety practices have improved significantly in modern times. Photo credit: Johan Hultin.
resize iconView Larger
A picture of Dr. Jeffery Taubenberger and Dr. Ann Reid reviewing a genetic sequence from the 1918 virus. They are credited with sequencing the genome of the 1918 virus. Photo Credit: National Museum of Health and Medicine Online Exhibit - MIS 377212.
resize iconView LargerIt wouldn’t be until 46 years later, in 1997, that Hultin would have another opportunity to pursue the 1918 virus. That year, Hultin came across an article in the journal Science authored by Jeffery Taubenberger et al. entitled, “Initial Genetic Characterization of the 1918 “Spanish” Influenza Virus.”6 At the time, Dr. Taubenberger was a young molecular pathologist working for the Armed Forces Institute of Pathology in Washington, D.C.
In the article, Taubenberger and his team described their initial work to sequence part of the genome of the 1918 virus. The genome is the complete list of genetic instructions that make up an organism, similar to a blueprint used for construction. Many people are familiar with the concept of DNA, which is dual-stranded and determines the fundamental genetic characteristics of nearly all living things. However, the genome of an influenza virus consists of single-stranded RNA instead. Taubenberger’s team team successfully extracted RNA of the 1918 virus from lung tissue obtained from a 21-year-old male U.S. service member stationed in Fort Jackson, South Carolina. The serviceman had been admitted to the camp’s hospital on September 20, 1918, with a diagnosis of influenza infection and pneumonia. He died six days later on September 26, 1918, and a sample of his lung tissue was collected and preserved for later study.
From this tissue, Taubenberger’s group was able to sequence nine fragments of viral RNA from four of the virus’ eight gene segments. This work did not represent a complete sequence of the entire 1918 virus’ genome, but it provided a clearer picture of the pandemic virus than ever before. Based on the 1918 virus’ sequence data Taubenberger assembled in 1997, he and his fellow researchers initially claimed that the 1918 virus was a novel influenza A (H1N1) virus that belonged to a subgroup of viruses that came from humans and pigs, as opposed to birds.6 However, there was still much to learn about the virus.
After reading Taubenberger’s article, Hultin once again became inspired to attempt to recover the 1918 virus. Hultin wrote a letter to Taubenberger, asking if Taubenberger would be interested if he could return to Brevig Mission and obtain lung tissues from victims of the 1918 virus buried in the Alaskan permafrost. During a return phone call, Taubenberger responded, yes. A week later, Hultin departed for Brevig Mission once again with meager tools for the task. He famously borrowed his wife’s garden shears to assist in the excavation.
Forty-six years had passed since Hultin’s first trip to the gravesite, and he was now 72 years old. He once again sought permission to excavate the gravesite from the village council — which he obtained — and he also hired locals to assist in the work. Hultin paid for the trip himself at a personal cost of about $3,200.7 The excavation took about five days, but this time Hultin made a remarkable find.
Buried and preserved by the permafrost about 7 feet deep was the body of an Inuit woman that Hultin named “Lucy.” Lucy, Hultin would learn, was an obese woman who likely died in her mid-20s due to complications from the 1918 virus. Her lungs were perfectly frozen and preserved in the Alaskan permafrost. Hultin removed them, placed them in preserving fluid, and later shipped them separately to Taubenberger and his fellow researchers, including Dr. Ann Reid, at the Armed Forces Institute of Pathology.5 Ten days later, Hultin received a call from the scientists to confirm — to perhaps everyone’s collective astonishment — that positive 1918 virus genetic material had indeed been obtained from Lucy’s lung tissue.
A picture of Johan Hultin at the Brevig Mission gravesite in 1997, 46 years after his first attempt to rescue the 1918 pandemic flu virus. Hultin saw that the small crosses that previously covered the site were missing, so Hultin built two large crosses (shown above) within the woodshop of a local school to mark the gravesite. Photo credit: Johan Hultin.
resize iconView Larger
Johan Hultin at age 72, during his second trip to the Brevig Mission burial ground in 1997. Photo credit: Johan Hultin.
resize iconView Larger
A picture of Johan Hultin excavating a body from the Brevig Mission burial ground. His wife’s garden shears, which Hultin borrowed to conduct the excavation, are shown in the center of the picture. Photo Credit: Johan Hultin.
Building the Blueprint
This is a picture of an influenza virus. Hemagglutinin (HA) is a surface protein of the virus that plays a role in allowing an influenza virus to enter and infect a healthy cell. Photo Credit: Dan Higgins, CDC.
The initial impact of this discovery would first be described in a February 1999 paper in the Proceedings of the National Academy of Science (PNAS) journal entitled “Origin and evolution of the 1918 “Spanish” influenza virus hemagglutinin gene,” by Ann Reid et al. 8 Hultin was acknowledged as a co-author. In the paper, the authors described their effort to sequence (i.e., characterize) the 1918 virus’s hemagglutinin “HA” gene.
The HA gene of an influenza virus determines the properties of the virus’s HA surface proteins. These HA surface proteins allow an influenza virus to enter and infect a healthy respiratory tract cell. HA is also targeted by antibodies produced by the immune system to fight infection. Modern flu vaccines work by targeting an influenza virus’ unique HA (a fact that virologist Dr. Peter Palese, featured later in this article, helped pioneer).
In the 1999 study, the authors succeeded in sequencing the full length HA gene sequence of the 1918 virus. To accomplish this, the authors used RNA fragments of the virus obtained from the bodies of the formerly described 21-year-old Fort Jackson service member, “Lucy” from Brevik Mission, and a third person, a 30-year-old male service member stationed at Camp Upton, New York. This man was admitted to the camp hospital with influenza on September 23, 1918, had a rapid clinical course of illness, and died from acute respiratory failure on September 26, 1918.
Sequencing results suggested that the ancestor of the 1918 virus infected humans sometime between 1900 and 1915. Drs. Reid and Taubenberger noted that the 1918 HA gene had a number of mammalian as opposed to avian adaptations, and was more human-like or swine-like depending on the method of analysis. Phylogenetic analysis, which is used to group influenza viruses in accordance with their evolutionary development and diversity, placed the 1918 virus’ HA within and around the root of the mammalian clade. This means that it likely was an ancestor or closely related to the earliest influenza viruses known to infect mammals. However, the authors believed the virus likely obtained its HA from avian viruses, but were unsure how long the virus may have been adapting in a mammalian host before emerging in pandemic form.
According to the authors, the existing strain to which the 1918 virus sequences were most closely related was “A/sw/Iowa/30,” the oldest classical swine influenza strain. The authors noted that contemporary avian influenza virus strains are very different from the 1918 pandemic virus, and unfortunately older avian strains from around the time of the 1918 pandemic were not available for study. The authors also noted that the 1918 virus’ HA1 had only four glycosylation sites, which is different from modern human HA’s which have accumulated up to five additional glycosylation sites through the process of antigenic drift. Antigenic drift refers to small changes in the genes of influenza viruses that happen continually over times as the virus copies itself. Antigenic drift is one reason why there is a flu season every year and also a reason for why people can get the flu multiple times in their lifetime.
Glycosylation sites are believed to be necessary for the function of influenza viruses, and the inclusion of additional glycosylation sites is believed to be an adaptation of the virus to human hosts. Also of note, the authors did not see any genetic changes in the 1918 virus’ HA that would explain its exceptional virulence.
Unlike modern virulent avian influenza strains, such as avian influenza A (H5) and (H7) viruses, the 1918 virus’ HA did not possess a “cleavage site” mutation, which is a recognized genetic marker for virulence, i.e., the severity or harmfulness of a disease. The insertion of amino acids in the HA cleavage site can allow an influenza virus to grow in tissues outside of its normal host cells. In the absence of such obvious markers, Dr. Reid and her fellow researchers concluded that there were likely multiple genetic factors responsible for the 1918 virus’ severity.
Microbiologist Dr. Peter Palese and his team created the plasmids used by Dr. Terrence Tumpey to reconstruct the 1918 pandemic virus. Palese has many accomplishments, including creating the first genetic maps of influenza A, B, and C viruses, as well as defining the mechanism used by the majority of current influenza antiviral drugs. Photo credit: Wikipedia (https://en.wikipedia.org/wiki/Peter_Palese)
Of note, the authors were able to sequence the entire code of the 1918 virus’ NA from the virus sample obtained from “Lucy’s” body. So here again, Hultin’s work proved invaluable. The authors found that the NA gene of the 1918 virus shared many sequence and structural characteristics with both mammalian and avian influenza virus strains.9 Phylogenetic analysis suggested the NA gene of the 1918 virus was intermediately located between mammals and birds, suggesting that it likely was introduced into mammals shortly before the 1918 pandemic. Furthermore, the 1918 virus’ NA obtained from Lucy suggested that it is very similar to the ancestor of all subsequent swine and human isolates.9A follow-up paper published in June 2000, entitled “Characterization of the 1918 “Spanish” Influenza Virus Neuraminidase Gene,” described sequencing of the 1918 virus’ neuraminidase (NA) gene.9 In an influenza virus, the neuraminidase gene is responsible for coding the virus’ NA surface proteins (see prior virus image for reference). An influenza virus’ NA surface proteins allow an influenza virus to escape an infected cell and infect other cells. Therefore, it plays an important role in spreading influenza infection. The author noted that NA is also targeted by the immune system, and that antibodies against NA do not prevent infection, but they do significantly limit the ability of the virus to spread.
Overall, the phylogenetic analysis seemed to indicate that the ultimate source of the 1918 virus’ NA was avian in nature, but the authors couldn’t determine the pathway from its avian source to the virus’ final pandemic form. Regarding genetic features of the NA that could explain the 1918 virus’ severity, the researchers were once again unable to find any single feature of the 1918 NA that contributed to the virus’ virulence.9 For example, in some modern influenza viruses, the loss of a glycosylation site in NA at amino acid 146 (in WSN/33) contributes to virulence and also results in the virus attacking the nervous system in mice. However, this change was not found in the NA of the 1918 virus.
Following this study, a series of additional studies were published, each detailing the findings from each of the 1918 virus’ remaining genes (flu viruses have 8 genes in total). In 2001, a paper by Christopher Basler et al. published in the Proceedings of the National Academic of Science (PNAS) described the sequencing of the 1918 virus’ nonstructural (NS) gene.10 A 2002 study in the Journal of Virology by Ann Reid et al. described sequencing of the virus’ matrix gene.11 Two years later, a 2004 Journal of Virology study described the sequencing of the 1918 virus’ nucleoprotein (NP) gene.12 In 2005, the virus’ polymerase genes were sequenced by Taubenberger et al and described in a Nature article.13 This final study bookended the nearly decade long process of sequencing the entire genome of the 1918 virus.
With the entire genome of the 1918 virus now sequenced, the necessary information was in place to reconstruct a live version of the 1918 virus. However, one more intermediate step was needed to start the reverse genetics process, which was to create plasmids for each of the 1918 virus’ eight gene segments.
This task was undertaken by renowned microbiologist, Dr. Peter Palese and Dr. Adolfo Garcia-Sastre at Mount Sinai School of Medicine in New York City. A plasmid is a small circular DNA strand that can be amplified (or replicated) in the laboratory. Years earlier, Dr. Palese helped pioneer the use of plasmids in reverse genetics to produce viable influenza viruses. The techniques he developed allowed the relationships between the structure and function of viral genes to be studied, and these efforts paved the way for the techniques used to reconstruct the 1918 virus. Once Dr. Palese and his colleagues at Mount Sinai completed creation of the plasmids, they were shipped to CDC so the official process of reconstruction could begin.
The Reconstruction
The decision to reconstruct the deadliest pandemic flu virus of the 20th century was made with considerable care and attention to safety. Senior government officials decided on CDC headquarters in Atlanta as the location of the reconstruction. CDC conducted two tiers of approvals: first by CDC’s Institutional Biosafety Committee and the second by CDC’s Institutional Animal Care and Use Committee, before work in the laboratory began. The work would be performed using stringent biosafety and biosecurity precautions and facilities, including what’s known as Biosecurity Level 3 (BSL-3) practices and facilities with enhancements.
A picture of Dr. Terrence Tumpey working in BSL3 enhanced laboratory conditions. This includes (but isn’t limited to) use of a powered air purifying respirator (PAPR), double gloves, suit, and working within a Class II biosafety cabinet (BSC). Today, Dr. Tumpey is the branch chief of the Immunology and Pathogenesis Branch in CDC’s Influenza Division. Photo credit: James Gathany - Public Health Image Library #7989.
resize iconView LargerFor reference, there are four biosafety levels that correspond to the degree of risk posed by research, with 1 posing the least risk and 4 posing the greatest risk. Each biosecurity level also corresponds with specific laboratory practices and techniques, personnel training requirements, laboratory equipment, and laboratory facilities that are appropriate for the operations being performed. The stringency of these considerations – again ranging from 1 as the lowest to 4 as the highest — is designed to protect the personnel performing the work, the environment and the community.
Each biosecurity level includes considerations for what is known as “primary” and “secondary” barriers. Examples of primary barriers include safety cabinets, isolation chambers, gloves and gowns, whereas secondary barriers include considerations such as the construction of the facility and HEPA filtration of air in the laboratory. The specific criteria for each biosafety level are detailed in the CDC/NIH publication Biosafety in Microbiological and Biomedical Laboratories.
A BSL3 laboratory with enhancements includes a number of primary and secondary barriers and other considerations. For example, all personnel must wear a powered air purifying respirator (PAPR), double gloves, scrubs, shoe covers and a surgical gown. They also must shower before exiting the laboratory. In addition, all work with the virus or animals must be conducted within a certified Class II biosafety cabinet (BSC), and airflow within the laboratory is directionally controlled and filtered so that it cannot accidentally exit the laboratory.
For the reconstruction of the 1918 virus, additional rules were created to govern the experiments to be conducted. For example, to prevent mix-ups and cross-contamination, work on the 1918 virus could not take place alongside work on other influenza viruses.
As part of security and safety considerations, CDC’s Office of the Director determined that only one person would be granted permission, laboratory access, and the tremendous responsibility of reconstructing the 1918 virus. That person was trained microbiologist Dr. Terrence Tumpey, who was approved for the project by then CDC director, Dr. Julie Gerberding. Reconstruction of the 1918 virus also was approved by the National Institute of Allergy and Infectious Disease (NIAID) within the National Institutes of Health (NIH), which partially funded the project.
Dr. Tumpey was formerly a U.S. Department of Agriculture microbiologist at the Southeast Poultry Research Laboratory in Athens, Georgia. Earlier in his career, he had applied for an American Society of Microbiology (ASM) postdoctoral fellowship with CDC microbiologist and flu expert Dr. Jacqueline Katz, who recently retired as Deputy Director of CDC’s Influenza Division. This two-year fellowship in CDC’s Influenza Division would mark the beginning of Dr. Tumpey’s career at CDC. He officially transferred employment to CDC for the purpose of studying human health implications of influenza viruses, including the 1918 pandemic virus.
The 1918 virus was extremely virulent. Image a) shows mouse lung tissue infected with a human seasonal H1N1 flu virus. Image c) shows the impact of the 1918 virus in mouse lung tissue. The 1918 virus replicates quickly and causes severe disease in the lung tissues of mice. In 1918, the virus caused severe disease in the lungs of people infected, as well. Photo credit: CDC, Science.
Using reverse genetics, Dr. Tumpey took the plasmids created by Dr. Palese for each of the 1918 virus’ eight gene segments and inserted them into human kidney cells. The plasmids then instructed the cells to reconstruct the RNA of the complete 1918 virus. For multiple weeks in July 2005, colleagues and collaborators asked Dr. Tumpey if he had the 1918 virus and if it had appeared in cell-culture yet.Dr. Tumpey’s work to reconstruct the complete 1918 virus began in the summer of 2005. To reduce risk to colleagues and the public, he was required to work on the virus alone and only after hours when fellow colleagues had exited the laboratories for the day and gone home. A biometric fingerprint scan was required for access into the BSL-3E laboratory, and the virus storage freezers were only accessible via an iris scan of his eyes. Dr. Tumpey was required to take a prescribed prophylactic (preventative) daily dose of the influenza antiviral drug, oseltamivir, as an additional safety precaution to prevent him from becoming infected. Should he become infected, he was informed that he would be placed in quarantine and denied contact with the outside world. He understood and accepted this responsibility and its consequences.
On the day the 1918 virus appeared in his cell-culture, Dr. Tumpey knew history had been made, and in fact, a historic virus had been brought back from extinction. He sent a playful, Neil Armstrong-inspired email later that day to colleagues and collaborators, which simply said “That’s one small step for man, one giant leap for mankind.” Everyone then knew what had been accomplished. Dr. Tumpey had become the first man to reconstruct the complete 1918 virus. The next step was to study it and unlock its deadly secrets.
Laboratory studies on the reconstructed 1918 virus began in August 2005. A report of this work, “Characterization of the Reconstructed 1918 Spanish Influenza Pandemic Virus”, was published in the October 7, 2005, issue of Science.14 To evaluate the 1918 virus’ pathogenicity (i.e., the ability of the virus to cause disease and harm a host), animal studies involving mice were conducted. The mice were infected with the 1918 virus, and measures of morbidity (i.e., weight loss, virus replication, and 50% lethal dose titers) were collected and documented. For comparison, other mice were infected with different influenza viruses that were designed via reverse genetics to have varying combinations of genes from the 1918 virus and contemporary human seasonal influenza A(H1N1) viruses. These viruses are called “recombinant viruses.”
The fully reconstructed 1918 virus was striking in terms of its ability to quickly replicate, i.e., make copies of itself and spread infection in the lungs of infected mice. For example, four days after infection, the amount of 1918 virus found in the lung tissue of infected mice was 39,000 times higher than that produced by one of the comparison recombinant flu viruses.14
The left picture shows replication of a human seasonal flu virus called Tx/91 in cell culture. The picture on the right shows how when the polymerase (PB1) gene of this same virus is exchanged with that of the 1918 virus, the resulting virus’ ability to replicate (i.e., make copies of itself) is greatly enhanced. Photo credit: Terrence Tumpey, CDC.
Furthermore, the 1918 virus was highly lethal in the mice. Some mice died within three days of infection with the 1918 virus, and the mice lost up to 13% of their body weight within two days of infection with the 1918 virus. The 1918 virus was at least 100 times more lethal than one of the other recombinant viruses tested.14 Experiments indicated that 1918 virus’ HA gene played a large role in its severity. When the HA gene of the 1918 virus was swapped with that of a contemporary human seasonal influenza A (H1N1) flu virus known as “A/Texas/36/91” or Tx/91 for short, and combined with the remaining seven genes of the 1918 virus, the resulting recombinant virus notably did not kill infected mice and did not result in significant weight loss.14
Other experiments were conducted to determine if infection with the 1918 virus could spread to other vital organs of mice — such as the brain, heart, liver and spleen. Laboratory testing did not detect virus in these organs, suggesting that the 1918 virus did not cause systemic infection in its victims.
However, one well-documented effect of the 1918 virus was rapid and severe lung damage. In 1918, victims of the pandemic virus experienced fluid-filled lungs, as well as severe pneumonia and lung tissue inflammation. Within four days post infection, mice infected with the 1918 virus experienced similar lung complications, suggesting that this was a unique aspect of the 1918 virus’ severity.14
The impact of the 1918 virus on lung tissue was also studied using a human lung cell line (known as Calu-3 cells). The amount of 1918 flu virus was measured in the cells at 12, 16 and 24 hours post infection and these results were compared to those produced by recombinant viruses with a combination of genes from the 1918 virus mixed with genes from contemporary human seasonal flu viruses. Similar to the experiments involving mice, the 1918 virus quickly multiplied and spread within the human lung cells. So much so, that the 1918 virus produced as much as 50 times the amount of virus in human lung cells as one of the comparison viruses. These experiments suggested that in addition to the HA, the polymerase genes of the 1918 virus played a significant role in the virus’ infectivity and virulence in human lung tissue.14
Another set of experiments was conducted to better understand the possible avian origins of the 1918 virus. The earlier sequencing efforts led by Dr. Taubenberger and Reid had suggested that the 1918 virus’ gene segments were more closely related to avian influenza A(H1N1) viruses than H1N1 viruses found in other mammals. Researchers were interested to know whether the 1918 virus would be lethal to fertilized chicken eggs, i.e., chicken eggs containing an embryo, similar to modern highly pathogenic avian influenza viruses.
To find an answer, 10-day old fertilized chicken eggs were inoculated with the 1918 virus. The 1918 virus proved lethal for the chicken egg embryos, similar to the effects caused by contemporary H1N1 bird flu viruses.14 Notably, comparison experiments using human seasonal influenza A(H1N1) viruses did not have this destructive effect on chicken embryos. Furthermore, the recombinant flu viruses that Dr. Tumpey created containing two, five or seven genes of the 1918 virus also did not hurt chicken embryos.14 Similar to the results of the studies conducted in mice and human lung cell, these fertilized chicken egg experiments indicated that the HA and polymerase genes of the 1918 virus both likely played roles in its virulence.
The work conducted by Dr. Tumpey and his CDC colleagues provided new information about the properties that contributed to the virulence of the 1918 virus. Dr. Tumpey determined that the HA and PB1 virus genes of the virus played particularly important roles in its infectiousness and severity. However, as his experiments involving recombinant flu viruses with some but not all of the 1918 virus’s genes showed, it was not any single component of the 1918 virus but instead the unique combination of all of its genes together that made it so particularly dangerous.
Tumpey and colleagues wrote “the constellation of all eight genes together make an exceptionally virulent virus.”14 No other human influenza viruses tested were as exceptionally virulent. In that way, the 1918 virus was special – a uniquely deadly product of nature, evolution and the intermingling of people and animals. It would serve as a portent of nature’s ability to produce future pandemics of varying public health concern and origin.
Top of Page
Learning from the Past
Since 1918, the world has experienced three additional pandemics, in 1957, 1968, and most recently in 2009. These subsequent pandemics were less severe and caused considerably lower mortality rates than the 1918 pandemic.2,3,4 The 1957 H2N2 pandemic and the 1968 H3N2 pandemic each resulted in an estimated 1 million global deaths, while the 2009 H1N1 pandemic resulted in fewer than 0.3 million deaths in its first year.3,4 This perhaps begs the question of whether a high severity pandemic on the scale of 1918 could occur in modern times.
Many experts think so. One virus in particular has garnered international attention and concern: the avian influenza A(H7N9) virus from China. The H7N9 virus has so far caused 1,568 human infections in China with a case-fatality proportion of about 39% since 2013. However, it has not gained the capability to spread quickly and efficiently between people. If it did, experts believe it could result in a pandemic with severity comparable to the 1918 pandemic. So far, it has shown only limited ability to spread between people. Most human infections with this virus have result from exposure to birds.
When considering the potential for a modern era high severity pandemic, it is important; however, to reflect on the considerable medical, scientific and societal advancements that have occurred since 1918, while recognizing that there are a number of ways that global preparations for the next pandemic still warrant improvement.
Besides the properties of the virus itself, many additional factors contributed to the virulence of the 1918 pandemic. In 1918, the world was still engaged in World War I. Movement and mobilization of troops placed large numbers of people in close contact and living spaces were overcrowded. Health services were limited, and up to 30% of U.S. physicians were deployed to military service.3
In addition, medical technology and countermeasures at the time were limited or non-existent. No diagnostic tests existed at the time that could test for influenza infection. In fact, doctors didn’t know influenza viruses existed. Many health experts at the time thought the 1918 pandemic was caused by a bacterium called “Pfeiffer’s bacillus,” which is now known as Haemophilus influenzae.
Influenza vaccines did not exist at the time, and even antibiotics had not been developed yet. For example, penicillin was not discovered until 1928. Likewise, no flu antiviral drugs were available. Critical care measures, such as intensive care support and mechanical ventilation also were not available in 1918.4 Without these medical countermeasures and treatment capabilities, doctors were left with few treatment options other than supportive care.3
In terms of national, state and local pandemic planning, no coordinated pandemic plans existed in 1918. Some cities managed to implement community mitigation measures, such as closing schools, banning public gatherings, and issuing isolation or quarantine orders, but the federal government had no centralized role in helping to plan or initiate these interventions during the 1918 pandemic.3
Today, considerable advancements have been made in the areas of health technology, disease surveillance, medical care, medicines and drugs, vaccines and pandemic planning. Flu vaccines are now produced and updated yearly, and yearly vaccination is recommended for everyone 6 months of age and older. Antiviral drugs now exist that treat flu illness, and in the event of virus exposure, can be used for prophylaxis (prevention), as well. Importantly, many different antibiotics are now available that can be used to treat secondary bacterial infections.
Diagnostic tests for identifying influenza are now available and they are improving over time. Current rapid tests for flu, also known as RIDTs, provide results within 15 minutes and have sensitivities ranging from 50-70%. Recently, new “rapid molecular assays” have become available that are timely and much more accurate than RIDTs. Just as important as these advancements in diagnostic tests are the improvements that have been made in laboratory testing capacity both within the United States and globally.
The World Health Organization (WHO)’s Global Influenza Surveillance and Response System (GISRS) is a global flu surveillance network that monitors changes in seasonal flu viruses and also monitors the emergence of novel (i.e., new in humans) flu viruses, many of which originate from animal populations. Through animal and human interactions and environmental exposures, these viruses can cause human infections. CDC in Atlanta is one of WHO’s six Collaborating Centers for Reference and Research on Influenza (joining others in Australia, China, Japan and the United Kingdom). The WHO collaborating centers collect influenza viruses obtained from respiratory specimens from patients around the world, and they are supported by 143 National Influenza Centers in 114 WHO member countries.3
Expanding laboratory testing and flu surveillance capacity around the world has been an important focus of pandemic preparedness efforts. In 2004, CDC began an international surveillance capacity building initiative that entailed a 5-year period of financial support to improve laboratory diagnostic tests and surveillance of influenza like illness (ILI) and severe acute respiratory infection (SARI) in 39 partner countries.
In 2008, CDC established the International Reagent Resource (IRR), which provides reagents to laboratories around the world to identify seasonal influenza A and B viruses, as well as novel influenza A viruses. During the 2009 H1N1 pandemic, the IRR distributed a new CDC developed 2009 H1N1 PCR assay to domestic public health laboratories and laboratories around the world less than 2 weeks after the 2009 H1N1 virus was first identified. This considerably enhanced the ability of the global flu surveillance community to track spread of the virus.3
As part of WHO’s International Health Regulations (IHR), countries must notify WHO within 24 hours of any case of human infection caused by a novel influenza A virus subtype. This requirement is designed to help quickly identify emerging viruses with pandemic potential.
Since 2010, CDC has used its Influenza Risk Assessment Tool (IRAT) to evaluate and score emerging novel influenza A viruses and other viruses of potential public health concern. The score provided by the IRAT answers two questions: 1) What is the risk that a virus that is novel in humans could result in sustained human to human transmission? and: 2) What is the potential for the virus to substantially impact public health if it does gain the ability to spread efficiently from person to person? Results from the IRAT have helped public health experts target pandemic preparedness resources against the greatest disease threats and to prioritize the selection of candidate vaccine viruses and the development of pre-pandemic vaccines against emergent viruses with the greatest potential to cause a severe pandemic.
When pre-pandemic vaccines are made, they are stored in the Strategic National Stockpile, along with facemasks, antiviral drugs and other materials that can be used in case of a pandemic.
All of these resources, tools, technologies, programs and activities are excellent tools for pandemic planning, and pandemic planning itself has improved significantly since 1918. In the United States, the Department of Health and Human Services (HHS) maintains a national Pandemic Influenza Plan, and this plan was updated in 2017. The World Health Organization (WHO) has published instructions for countries to use in developing their own national pandemic plans, as well as a checklist for pandemic influenza risk and impact management.3
Planners have access to other materials as well. For example, in 2014, CDC published a pandemic framework with six intervals that fall within a pandemic curve. Each interval helps with prioritizing data collection, government resources and interventions, and other important activities during the pandemic. In addition, CDC experts have devised a Pandemic Severity Assessment Framework that uses data to assign severity and transmissibility scores to pandemics. The tool is useful for planning purposes and for determining appropriate mitigations based on the severity of a pandemic. In addition, guidelines for non-pharmaceutical interventions, such as closing schools and large social gatherings, have been established and revised, for use during a pandemic.
While all of these plans, resources, products and improvements show that significant progress has been made since 1918, gaps remain, and a severe pandemic could still be devastating to populations globally. In 1918, the world population was 1.8 billion people. One hundred years later, the world population has grown to 7.6 billion people in 2018.3 As human populations have risen, so have swine and poultry populations as a means to feed them. This expanded number of hosts provides increased opportunities for novel influenza viruses from birds and pigs to spread, evolve and infect people. Global movement of people and goods also has increased, allowing the latest disease threat to be an international plane flight away. Due to the mobility and expansion of human populations, even once exotic pathogens, like Ebola, which previously affected only people living in remote villages of the African jungle, now have managed to find their way into urban areas, causing large outbreaks.
If a severe pandemic, such as occurred in 1918 happened today, it would still likely overwhelm health care infrastructure, both in the United States and across the world. Hospitals and doctors’ offices would struggle to meet demand from the number of patients requiring care. Such an event would require significant increases in the manufacture, distribution and supply of medications, products and life-saving medical equipment, such as mechanical ventilators. Businesses and schools would struggle to function, and even basic services like trash pickup and waste removal could be impacted.
The best defense against the flu continues to be a flu vaccine, but even today, flu vaccines face a number of challenges. One challenge is that flu vaccines are often moderately effective, even when well matched to circulating viruses. But perhaps the biggest challenge is the time required to manufacture a new vaccine against an emerging pandemic threat. Generally, it has taken about 20 weeks to select and manufacture a new vaccine.
During the 2009 H1N1 pandemic, the first doses of pandemic vaccine did not become available until 26 weeks after the decision to manufacture a monovalent vaccine.3 As a result, most vaccinations in the United States occurred after the peak of 2009 H1N1 illness. The HHS Pandemic Influenza Plan has a goal of reducing the timeframe to make a pandemic flu vaccine from 20 weeks to 12 weeks, but accomplishing this is challenging.
One possible solution is to create more broadly protective and longer lasting vaccines. Creation of a “universal vaccine” continues to elude the world’s top scientists, but in the future, it could become a reality. In the meantime, health officials seek to get the most out of new and existing flu vaccine technologies, such as cell based and recombinant vaccines, which are not reliant on a supply of chicken eggs, like traditional vaccines, and have the potential to be produced faster.
One other vaccine issue is the inadequate global capacity for mass producing flu vaccines. Global pandemic flu vaccine capacity was estimated to be 6.4 billion doses in 2015, but this is not enough to cover even half of the world’s population, should two doses of a pandemic vaccine be required for protection.3
Other challenges at a global level include surveillance capacity, infrastructure and pandemic planning. The majority of counties that report to the WHO still do not have a national pandemic plan, and critical and clinical care capacity, especially in low income countries, continues to be inadequate to the demands of a severe pandemic.3 In 2005, milestones were created in the revised International Health Regulations (IHR) for countries to improve their response capacity for public health emergencies, but in 2016, only one-third of countries were in compliance.3
All of these issues show that more work needs to be done, both here in the United States and internationally, to prepare for the next pandemic. On May 7, 2018, The Rollins School of Public Health at Emory University in partnership with the U.S. Centers for Disease Control and Prevention, hosted a one-day symposium on the 100-year anniversary of the 1918 influenza pandemic. The event involved experts from government and academia discussing current pandemic threats and the future of pandemic preparedness, influenza prevention and control. U.S. and global influenza experts who attended the meeting agreed that we still face great challenges to prepare for future flu pandemics, but part of the solution is recognizing these challenges and working together with the rest of the world to address them.
For more information on the 1918 pandemic, see CDC’s 1918 (H1N1 virus) website. For more information about influenza pandemics, see Pandemic Influenza.
GIA', hanno resuscitato l'influenza spagnola. E io che pensavo che avrebbero clonato i MAMMUTH, bah... chissà adesso dove e perché questi preziosi virus sono conservati, a cosa serviranno mai se l'epidemia era estinta naturalmente?